|
Ingesting or inhaling calcium metal can be fatal due to the burns it can produce. Skin contact with calcium metal can cause irritation, corrosion, and chemical burns. Calcium metal reacts exothermically with water or acid.People report is as contributing a mineral, sour, or salty flavor. People and other animals can often taste the calcium ion.The nearest noble configuration is 1s22s22p63s23p6. Calcium metal is soft enough to cut with a knife, although somewhat harder than the metal lead. Therefore the electronic configuration of calcium is 1s22s22p63s23p64s2.Calcium salts are used to produce orange in fireworks. Calcium is used in fireworks to deepen the color. Calcium X-ray photoelectron spectra, calcium electron configuration, and other elemental.And the electron configuration of calcium is 1s2 2s2 2p6 3s2 3p6 4s2. 3: For sodium it is better to use the convention of expressing the core electrons with that of a Nobel gas. Hence the electron arrangement in calcium is 2, 8, 8, 2. That is, you could write Na: 1s 2 2s 2 2p 6 3s 1 or Ne3s 1 noting that Ne1s 2 2s 2 2p 6. Calcium burns with a dark red color in a flame test. Now the atomic number of calcium (Ca) is 20.Approximately one third of the mass of the human body is calcium after all water is removed. Calcium is the fifth most abundant element in the human body.However, too much calcium can lead to kidney stones or artery calcification. Calcium is the main component of teeth and bones.The top three countries that produce calcium are China, United States and India.Some of the most common compounds found on Earth are limestone (calcium carbonate - CaCO 3), gypsum (calcium sulfate - CaSO 4♲H 2O), fluorite (calcium fluoride - CaF 2) and apatite (calcium fluorophosphate - CaFO 3P or calcium chlorophosphate - CaClO 3P) The electron configuration of a neutral calcium atom is '1s'2'2s'2'2p'6'3s'2'3p'6'4s'2'. A neutral calcium atom also has 20 electrons. Calcium is not found free in nature, but calcium compounds are common. This means that in a neutral calcium atom, there are 20 protons in its nucleus.Calcium is the 5th most abundant element in the Earth's crust, making up 3.22% of the earth, air, and oceans.configuration Ne3s1 of elements A, B and Care He2s1, and Ar4s1. Mårtensson, "Core-Level Binding Energies in Metals," J. Ca: Color: dull gray or silver: Atomic Number: 20: Category: alkaline earth metal: Atomic Weight: 40. electron will break this stable electronic configuration and requires high. Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000. Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978. Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5). The data are adapted from references 1-3. I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. The binding energies are quoted relative to the vacuum level for rare gases and H 2, N 2, O 2, F 2, and Cl 2 molecules relative to the Fermi level for metals and relative to the top of the valence band for semiconductors. The electronic configurations of the elements in group 1: The atoms of all group 1 elements have similar chemical properties and reactions because they all have one electron in their outer shell. 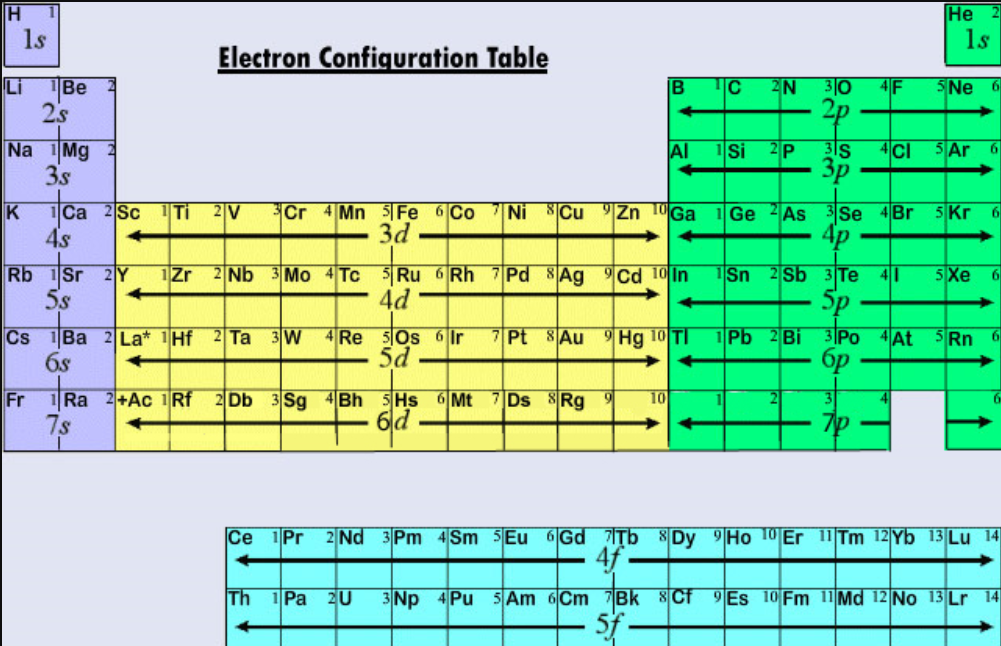
Calcium 1 s 2 2 s 2 2 p 6 3 s 2 3 p 6 4 s 2. Each elements electron configuration, which was determined in the previous section, is shown below. All values of electron binding energies are given in eV. Determine how many of the electrons in each of the following elements are classified as valence electrons. electron dot structure for the formation of this oxide. 1967, 47, 1300.Įlectron binding energies Electron binding energies for calcium. element with variable ( more than one ) valency stored under water. 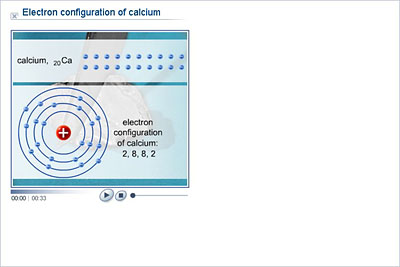
These effective nuclear charges, Z eff, are adapted from the following references:
0 Comments

My preference is to have a single transformer provide power to two wreaths (therefore, powering four separate 20 LED strings), but I also could see using one transformer for each wreath (power to just two separate 20 LED strings), if that's simpler. I have an outside outlet I could use, but I'm concerned that available transformers might not be weatherproof, so it seems better to me to keep the transformers inside the house. I envision plugging in the transformers inside and running the DC power wires out through the open windows, then closing the windows down on the wires for the season. These wreaths hang outside on four windows, two of those windows are close to each other on each side of the front of the house. And if I run more than one string from a transformer, would I need to multiply the required minimum amperage by the number of strings being powered to know the actual minimum amperage necessary to run that number of strings? So each wreath has two (2) battery boxes, and therefore each wreath requires 4 "D" cell batteries.įrom this, I'm guessing that each string requires 3 volts, but I have no idea what amperage. Each string is powered by two (2) "D" cell batteries in a box. I assume that, with that information, I can buy the correct transformers at Frys or on Amazon.Įach of these wreaths has two (2) strings of 20 LED white lights. But I'm not sure what capacity transformer to get and how to wire it up correctly.Ĭan someone here can tell me the voltage and minimum amperage each transformer will require and how to be sure I wire it correctly?.  
I want to convert them to run from an AC/DC transformer, because dealing with replacing all these batteries in these each year is such a PITA that we just don't want to do it again (8 tiny screws per each of 8 battery boxes!). We have four (outdoor) Xmas wreaths with white LED lights that are battery operated. 
Coaching cues will not change a players fundamental play styles.Įven though NFL Head Coach uses Madden 06's engine, it's heavily modified. In real life, we say the QB didn't trust his arm to make the throw. This happened because, even though the QB could be 85+ overall, the scheme plays to his weakness with arm strength. You tell him "Stay in the pocket", and then "Wait for the play to develop" for even more emphasis on making him throw instead of scrambling. He keeps scrambling despite being only a moderately quick QB. You have a noodle arm QB in a "SPREAD" system. The issue is the game doesn't tell you there's an issue. The AI isn't great that's for sure, but there's a reason for everything that happens in either of the games. EA did a terrible job promoting both of them, and left way too many features to be swept under the rug. This is a major reason why the HC franchise died, people didn't know how to play the game and when they were losing they thought the game was broken. WHAT DO YOU MEAN I NEED TO HOW LEARN TO PLAY NFL HEAD COACH? I haven't been able to find the values of how many yards each coach expects to earn/allow, if i find them I'll make a separate post on here.) Your defensive staff will be impossible to retain, and your offensive coaches will get their attributes cheesed. ( NOTE: It's not recommended to change the QTR length because it exploits the total yards per game. This program allows you to adjust everything Madden would, with a few bonuses like "sim injuries" and QTR length. NFL HOF QB's (like Peyton Manning) will routinely throw 5INTs against a offensive ROOKIE Head Coach and the road to becoming the greatest coach of all time is easily handed to you within your first 5 seasons. The AI is way way too broken to be played without sliders. The reason for this is that there's no sliders on console. Only the legitimate PC version of the game. One of the major things about NFL Head Coach is that it's only playable on PC. And finally, I created the r/NFLHeadCoachSeries subreddit that is the largest active HC community there is. I learned how to edit the game from within while learning about all the factors that come into play while doing so. Multiple careers, schemes, players, coaches, etc. I've played NFL Head Coach for a few years, religiously. This will be a guide on learning how to play the game, rather than learning how to win/succeed in the game.įirst lets start with my background on it. Moved it to a public S3 bucket that will not expire hereĮdit (): The latest build of RPCS3 causes the game to crash out when you select the first play of a game in career mode.NFL Head Coach: The game that requires you to learn how to play it, while learning to play it. I’ll drop it into a new location soon when I get a chanceĮdit (again ): The config file in gofile.io must have expired. I've simulated an entire season, and coached a few games with no crashes since I got the game running in a stable stateĮdit: forgot to mention I’m running a build of RPCS3 from yesterday (11th Feb 2021)Įdit (): looks like the config file I dropped into go file.io timed out. Drop this Config file into your RPCS3 folder at config/custom_configs, and it should be picked up by the BLUS30128 version of NFLHC09įor me, this config boots into the game, and it runs flawlessly, EXCEPT one issue where the field looks dirty, but it's something I'm willing to ignore for a working version of the game. 
In RPCS3, download v1.02 of the game from here.Windows 10, latest updates, latest NVidia drivers.I spent the guts of 4-5 hours testing different configurations on my machine Posting my configuration in case it helps anyone else here. I've seen a few posts about this game not working on RPCS3, common issues from googling are |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
